Answers
Explanation:
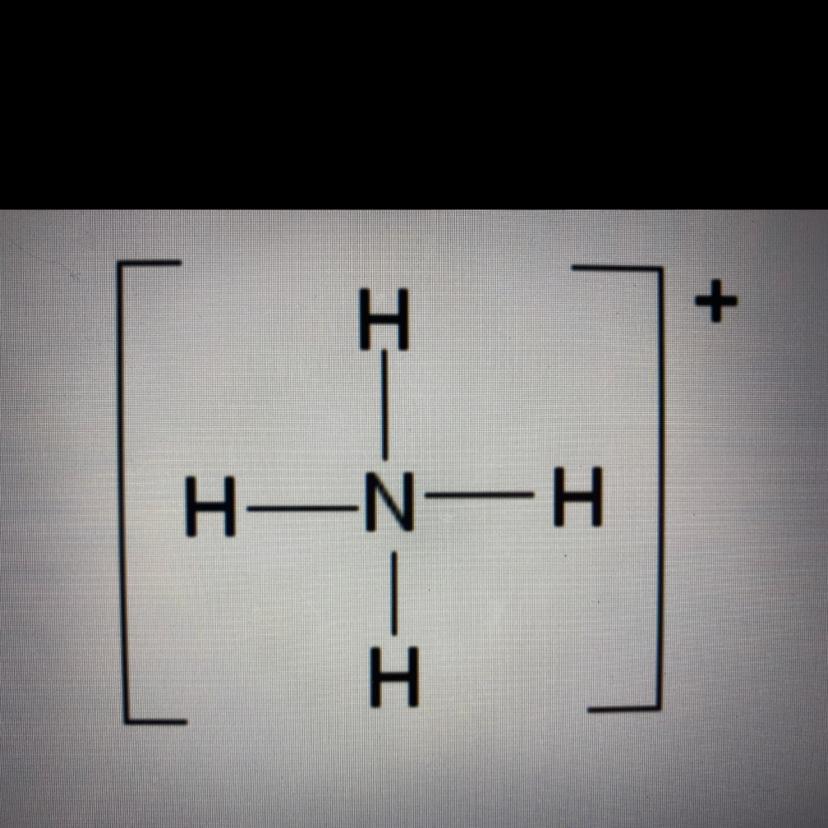
As I think Option C is correct i.e. NH4+.
Related Questions
How many valance electrons does He need to get to 8
Answers
Answer:
Any element in group 18 has eight valence electrons (except for helium, which has a total of just two electrons
A cylinder of gas with a pressure of 8.0 atm is heated from 300. K to 600. K. What will the new pressure of the gas be?
Answers
Answer:
[tex]P_2=16atm[/tex]
Explanation:
Hello there!
In this case, according to the given information, we can infer we need to use the Gay-Lussac's equation it order to understand this pressure-temperature relationship as shown below:
[tex]\frac{P_1}{V_1}=\frac{P_2}{V_2}[/tex]
Thus, we solve for P2 and plug in P1, T1 and T2, to obtain:
[tex]P_2=\frac{P_1T_2}{T_1}\\\\P_2=\frac{8atm*600K}{300K}\\\\P_2=16atm[/tex]
Regards!
does the moon really change shape? why
Answers
What type of bond will be formed between Ar and Cu
Answers
Find the % composition for each element in Zinc Chlorate
Answers
Answer:
chlorine ~ 30%
zinc ~ 28%
oxygen ~ 41%
Explanation:
is this correct im just asking because my little brother not sure for his answer
Answers
7
State two signification of melting?
Answers
For the following reaction conditions select the correct statement regarding the reaction below.
OH Cl OTs OTs N TsCl W X Y Z N
a. W is the correct product.
b. X is the correct product.
c. Y is the correct product.
d. Z is the correct product since pyridine acts as a nucleophile.
e. Both X and Y are both formed in this reaction as a racemic mixture
Answers
Answer:
B
Explanation:
The appropriate diagram of the question is shown in the first image attached below.
From the diagram, we see the reaction of Cyclopentanol taking place under Tscl pyridine. We are to show the reaction mechanism and determine from the options, which appropriate product fits in.
So, from the reaction, the hydroxyl substituent reacts with Tscl where cl is being lost. This process is followed by an attack of N substituent on the pyridine with the Hydrogen atom and cleaves off for the structure to form a stable structure. The stereochemistry of the compound remains unchanged and it maintains its stick formula.
Thus, X is the appropriate and the correct product.
Can someone plss help me answer those 4 questions by tonight.Thank you !
Answers
Answer:
no problem hey I will help you
The total pressure in a tank is filled with a mixture of gases : Oxygen, Helium, and Argon is 15.3 atm. The P(He) = 3.2 atm, P(O) = 7.4 atm, what is the P(Ar) in the tank?
Answers
Answer:
4.7 atm
Explanation:
From the question given above, the following data were obtained:
Total pressure (P) = 15.3 atm
Pressure of Helium, P(He) = 3.2 atm
Pressure of Oxygen, P(O) = 7.4 atm
Pressure of Argon, P(Ar) =?
The pressure of Argon, P(Ar) in the tank can be obtained as illustrated below:
P = P(He) + P(O) + P(Ar)
15.3 = 3.2 + 7.4 + P(Ar)
15.3 = 10.6 + P(Ar)
Collect like terms
15.3 – 10.6 = P(Ar)
4.7 = P(Ar)
P(Ar) = 4.7 atm
Therefore, the pressure of Argon, P(Ar) in the tank is 4.7 atm
pls help i need asap will mark brainlest
Answers
Answer:
B. 50%
Explanation:
2H₂ + CO → CH₃OHFirst we convert the given masses of the reactants into moles, using their respective molar masses:
4 g H₂ ÷ 2 g/mol = 2 mol H₂25 g CO ÷ 28 g/mol = 0.893 mol CO0.893 moles of CO would react completely with (0.893 * 2) 1.786 moles of H₂. As there are more H₂ moles than that, H₂ is the reactant in excess and CO is the limiting reactant.
Now we calculate how many CH₃OH moles would have been formed if all CO would have been consumed:
0.893 mol CO * [tex]\frac{1molCH_3OH}{1molCO}[/tex] = 0.893 mol CH₃OHThen we convert 0.893 moles of CH₃OH into grams, using its molar mass:
0.893 mol CH₃OH * 32 g/mol = 28.57 gFinally we calculate the percent yield:
14 g / 28.57 g * 100% = 49%Calculate the freezing point of a solution made by dissolving 13 g potassium sulfide in 150 g H2O Kf for water = 1.86 C/m
Answers
Answer:
[tex]T_F=-4.4\°C[/tex]
Explanation:
Hello there!
In this case, according to the given information, it is possible for us to calculate the freezing point of an aqueous solution of potassium sulfide by using the following equation:
[tex]T_F=T_{solv}-i*m*Kf[/tex]
In such a way, we firstly calculate the molality of this solution according to:
[tex]m=\frac{\frac{13g}{110.262 g/mol} }{0.150kg} =0.79m[/tex]
Finally, since the Van't Hoff's factor for K2S is 3, the freezing point of the solution turns out to be:
[tex]T_F=0\°C-3*0.79m*1.86\°C/m\\\\T_F=-4.4\°C[/tex]
Regards!
SOMEONE HELP ME
1. How many grams of C are present in 4.86 grams of carbon dioxide
?
grams C.
2. How many grams of carbon dioxide contain 1.73 grams of O ?
grams carbon dioxide.
Answers
Answer:
1. 1.33 gram of carbon
2. 2.38g of carbon dioxide
Explanation:
From the given information:
Total amount of CO₂ = 4.86 grams
Atomic mass of C = 12 g/mole
molar mass of CO₂ = 44 g/mole
∴
The mass of the Carbon (C) in grams is:
[tex]= Total\ amount \ of \ CO_2 \times \dfrac{12 \ g/mol}{44 \ g/mol}[/tex]
[tex]= 4.86 \ g \times \dfrac{12 \ g/mol}{44 \ g/mol}[/tex]
= 1.33 gram of carbon
2.
Here, the total amount of CO₂ = unknown
Atomic mass of O₂ = 32 g/mole
molar mass of CO₂ = 44 g/mole
amount of oxygen = 1.73 g
∴
The mass of CO₂ = [tex]Total \ amount \ of \ O_2 \times \dfrac{44 \ g/mol}{32\ g/mol}[/tex]
[tex]=1.73 \times \dfrac{44 \ g/mol}{32\ g/mol}[/tex]
= 2.38 g of carbon dioxide
Control rods in nuclear reactors are made of materials that absorb free neutrons in order to
slow down the chain reaction.
True
False
Answers
If a particular ore contains 56.3 % % calcium phosphate, what minimum mass of the ore must be processed to obtain 1.00 kg k g of phosphorus?
Answers
Answer:
34.44 kg
Explanation:
First we convert 1.00 kg of phosphorus (P) into moles, using its molar mass:
1.00 kg ÷ 32 kg/kmol = 0.03125 kmol PThen we convert 0.03125 kmoles of P into kmoles of Ca₃(PO₄)₂:
0.03125 kmol P * [tex]\frac{2kmolCa_3(PO_4)_2}{2kmolP}[/tex] = 0.0625 kmol Ca₃(PO₄)₂Now we calculate the mass of 0.0625 kmoles of Ca₃(PO₄)₂:
0.0625 kmol Ca₃(PO₄)₂ * 310.18 kg/kmol = 19.39 kgFinally we calculate the required mass of the ore, using the definition of content percentage:
% content = Mass of calcium phosphate / mass of ore * 100 %56.3 % = 19.39 kg / mass of ore * 100%Mass of Ore = 34.44 kgPlease help will give brainliest
Perform the following
mathematical operation, and
report the answer to the correct
number of significant figures.
328 x 0.125 = [?]
Answers
Answer: 41.0
Explanation: When you multiply the two numbers you get 41 but you need to have the same amount of significant numbers as the number in the problem with the least significant numbers. I hope this helps
Can someone give me an example of balancing equations with a solution that is simple?
Answers
Explanation:
[tex]H _{2}O _{2(aq)} →H _{2}O _{(l)} + O _{2}(g) \\ solution : 2 \: and\: 2[/tex]
The equation:
K + HOH -> KOH + H2 (an example of a single replacement reaction)
Balanced equation:
2 K + 2 HOH -> 2 KOH + 1 H2
In order to use the Ideal Gas Constant of 0.0821, what units must be used for volume, pressure, amount, and temperature?
Answers
Answer:
The value becomes 0.0821 L-atm/K⁻¹-mol⁻¹.
Explanation:
The value of gas constant is 0.0821.
We know that, the ideal gas law is as follows :
[tex]PV=nRT\\\\R=\dfrac{PV}{nT}[/tex]
Where
P is pressure
V is volume
T is temperature
In order to use the Ideal Gas Constant of 0.0821, the units are follows :
Volume = Litre (L)
Pressure = atm
Amount = mol⁻¹
Temperature = K⁻¹
So, the value becomes 0.0821 L-atm/K⁻¹-mol⁻¹.
If you have a solution that is 15 percent by mass of KCl in benzene, what is the new boiling point?
Answers
Answer:
https://www.chegg.com/homework-help/questions-and-answers/1-10-points-solution-15-percent-mass-kcl-benzene-new-boiling-point--901-c-b-921-c-c-821-c--q63751186
Explanation: Thats your answer
Colligative properties of solutions include all of the following except: a. an increase in the osmotic pressure of a solution upon the addition of more solute b. elevation of the boiling point of a solution upon addition of a solute to a solvent c. an increase of reaction rate with increase in temperature d. depression of the freezing pont of a solution upon addition of a solute to a solvent e. depression of vapor pressure upon addition of a solute to a solvent
Answers
Answer:
Option C, an increase of reaction rate with increase in temperature
Explanation:
Colligative properties are as follows
a) Decrease of vapor pressure
b) Increase of boiling point
c) Reduction of freezing point
d) Increase of osmotic pressure
There is no impact on reaction rate and hence it is not a colligative property.
Thus, option c is the right choice
An increase of reaction rate with increase in temperature isn't an example
of colligative properties of solutions
Colligative properties of solutions depend on the ratio of the number of
solutes to that of the solvent(concentration) and not on the nature of the
substances involved.
Examples of colligative properties include vapor pressure lowering, boiling
point elevation, freezing point depression, and osmotic pressure. Increase of
reaction rate with increase in temperature is therefore not an example of
colligative properties of solutions.
Read more on https://brainly.com/question/24260365
. Explain why some desert animals excrete uric acid rather than ammonia.
(2 marks)
Answers
Answer:
AFAIK
Explanation:
uric acid is much less toxic than ammonia, hence bigger concentrations of it are tolerated in the body. This means you can excrete it while excreting very little water - beneficial wherever water's not abundant.
There's a tradeoff though, uric acid requires more energy to synthesize than ammonia, so pretty much all fish, say, excrete ammonia rather than uric acid - it's no problem to dilute ammonia since there's no water shortage.
During a combustion reaction, 9.00 grams of oxygen reacted with 3.00 grams of CH4.
What is the amount of the leftover reactant?
0.74 grams of methane
0.89 grams of methane
1.22 grams of oxygen
1.45 grams of oxygen
Answers
Answer:d
Explanation:
A solution at 25 degrees Celsius has a pH of 4.48. What is the pOH of this solution?
0.978
3.17
9.52
10.51
Answers
Answer:
The answer is C = 9.52
Explanation:
I took the test
The pOH of a solution that has a pH of 4.48 at 25°C is 9.52. Details about pH can be found below.
What is pH?pH refers to the power of hydrogen in a substance while the pOH refers to the power of hydroxyll ions. The pH of a solution ranges from 0 - 14 indicating the strength of acidity and alkalinity.
The relationship between the pH and pOH in a solution is given as follows:
pH + pOH = 14
According to this question, a solution at 25 degrees celsius has a pH of 4.48. The pOH of the solution is calculated as follows:
4.48 + pOH = 14
pOH = 14 - 4.48
pOH = 9.52
Therefore, the pOH of a solution that has a pH of 4.48 at 25°C is 9.52.
Learn more about pH at: https://brainly.com/question/15289741
Use the nutrition label to answer the following questions.
16.How much energy is contained in
the six-cookie serving size
recommended on the label?
17.How much energy in joules is provided by eating
six cookies? (1 cal = 4.184 J; ALSO: 1 Calorie = 1
kilocalorie)
Answers
Answer:
six cookies?(1 cal _4.184 j; ALSO: 1 Calorie -1 kilocalorie)
how to make 100 ml of 0.001 mM solution with 0.0405mM solution?
Answers
Answer:
Measure 2.47 mL of the stock solution (i.e 0.0405 mM) and dilute it to the 100 mL mark with water
Explanation:
To make 100 mL of 0.001 mM solution from 0.0405mM solution, we need to determine the volume of 0.0405mM solution needed. This can be obtained as follow:
Molarity of stock (M₁) = 0.0405 mM
Volume of diluted (V₂) = 100 mL
Molarity of diluted solution (M₂) = 0.001 mM
Volume of stock solution needed (V₁) =?
M₁V₁ = M₂V₂
0.0405 × V₁ = 0.001 × 100
0.0405 × V₁ = 0.1
Divide both side by 0.0405
V₁ = 0.1 / 0.0405
V₁ = 2.47 mL
Therefore, to make 100 mL of 0.001 mM solution from 0.0405mM solution, measure 2.47 mL of the stock solution (i.e 0.0405 mM) and dilute it to the 100 mL mark with water.
______ is the process of change from a liquid to a gas at temperatures below the boiling
point.
Answers
Answer:
evaporation is the process
Answer:
EvaporationExplanation:
Evaporation is the process of becoming a vapor. The process of extracting moisture as by heat.
Evaporation is the process of change from a liquid to a gas at temperatures below the boiling point.
Therefore, the final answer is evaporation.
Use the scenario to answer the following question. A group of students working in a chemistry lab are planning a procedure to neutralize 10.0mL of 5.0 M hydrochloric acid (strong acid) with 5.0 M potassium hydroxide (strong base). In their procedure they plan on adding an equal volume of the base to the acid. What would be the expected outcome of carrying out this step as planned?
a- The resulting pH will be less than 7 because potassium hydroxide is less concentrated than the hydrochloric acid.
b- The resulting pH will not be able to be determined because the concentrations of the acid and base are not the same.
c- The resulting pH will be greater than 7 because potassium hydroxide is less concentrated than the hydrochloric acid.
d- The resulting pH will be equal to 7 because a strong base will neutralize a strong acid.
Answers
Answer:
d- The resulting pH will be equal to 7 because a strong base will neutralize a strong acid.
Explanation:
The reaction between potassium hydroxide and hydrochloric acid of equal volume and equal concentration yields a solution of pH 7 at equivalence point. We must note that KOH is a strong base while HCl is a strong acid. This fact influences the pH of the system at equivalence point.
Owing to the fact that the acid is exactly neutralized by the base; at the equivalence point of such titration, it is expected that hydrogen ions(H+) and hydroxide ions (OH-) must have reacted to form water, this leads to a final pH of 7.
Hydrogen iodide can decompose into hydrogen and iodine gases. 2HI(g) H2(g) I2(g) K for the reaction is 0.016. Of 0.148 atm of HI(g) is sealed in a flask, what is the pressure of each gas when equilibrium is established
Answers
Solution :
Given :
Hydrogen iodide decomposes to hydrogen and iodine gas
[tex]$2 HI \ \ \ \Leftrightarrow \ \ \ \ H_2 \ \ \\ + \ \ I_2 $[/tex]
I 0.148 0 0
C -2a +a +a
E 0.148-2a a a
We know
[tex]$k_p=\frac{P(H_2)P(I_2)}{P(HI)^2}$[/tex]
[tex]$0.016=\frac{a^2}{(0.148-2a)^2}$[/tex]
[tex]$0.016^{1/2}=\frac{a}{0.148-2a}$[/tex]
[tex]$0.12649=\frac{a}{0.148-2a}$[/tex]
0.0187 = 1.2529 a
a = 0.0149
Therefore
P(HI) = 0.148 - 2a
= 0.148 - 2(0.0149)
= 0.1182 atm
P([tex]$H_2$[/tex]) = a
= 0.0149 atm
P([tex]$I_2$[/tex]) = a
= 0.0149 atm
what is the major organic product obtained from the following sequence of reactions 1. naoch2ch3 ch3ch2oh 2. ph br 1. naoch2ch3 ch3ch2oh 2. br 1. lialh4 et2o 2. h3o
Answers
Which chemical equation is balanced as written?
PLS ANSWER FAST
Answers
Answer:
THERE ANSWER IS C
Explanation: